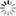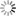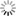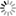What are OLEDs and OLETs?
OLED (Organic Light-Emitting Diode)
An organic light-emitting diode (OLED) is a solid-state device whose emissive electroluminescent layer comprises a film of organic material sandwiched between two electrodes. OLEDs can be based on thin films of organic small molecules or polymers that generate light when a voltage bias is applied between electrodes. Usually the two electrode materials are selected to favour hole (anode) and electron (cathode) injection. Generally the anode is made of ITO (Indium-Tin-Oxide, transparent material with high work function, ideal for hole injection) and the cathode of Al (low work function material, ideal for electron injection). The first demonstration of the OLED dates back to the 1960s when electrically driven light emission from non-crystalline organic materials was first observed [1]. After that, several studies were carried out by academic groups and companies (Kodak, Pioneer, Motorola, NEC, etc…) both for fundamental physics comprehension and application purposes. The study of the OLEDs was so impressive and massive because they are, combined with transistors, the main component for flat panel displays. Nowadays electronic products containing displays are becoming more and more portable. Therefore, they need some peculiarities like lightweightness, flexibility, brightness, etc… These, with many others, are the strong points of the OLEDs. In fact they are thinner, lighter and more flexible with respect to their inorganic counterpart. Moreover, OLEDs can be as bright as LEDs and they consume much less power. Due to the organic processabilty, they are easier to produce and can be made to larger area. Finally OLEDs have large fields of view, about 170 degrees, a significantly advantage over, for example, liquid crystal displays. Obviously, these devices present also some disadvantages: they have typically shorter lifetime (in particular life time of the blue emitter is critical, about 1.000 hours), they are not very stable and can easily be contaminated by water or oxygen. Most of organic materials (also the most highly emitting) show either p-type (hole) or ntype (electron) charge transport characteristics [2, 3]. For this reason different organic materials are combined in a multi-layer architecture to improve the performances. It can thus be employed a material for hole transport, another one for electron transport and a third one for light emission (Fig. 1).
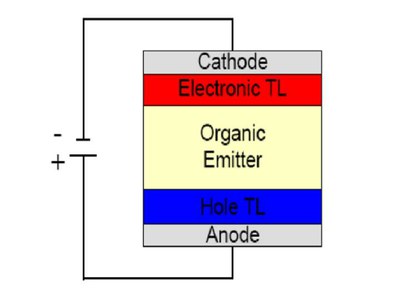
Figure 1: Scheme of a classic 3-layer OLED architecture
To understand the basic working principles of an OLED we can use the example of a basic three-layer device reported in Fig. 1. When a positive electrical potential is applied to the anode, the injection of holes occurs from this electrode into the hole transport layer (HTL), while the injection of electrons occurs from the cathode to the electron transport layer (ETL). The injected carriers move through the organic materials toward the oppositely charged electrode.
The OLED works if the energy gap and the energetic positions of the HOMO and LUMO levels of the materials are positioned as in the scheme of Fig. 2. The hole injection from the HTL layer to the organic emitter is supported, ensuring hole blocking at the ETL one.
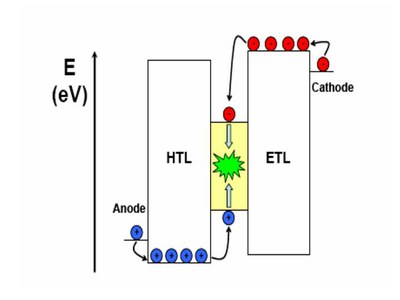
Figure 2. Scheme of energy levels for a three layers OLED. On the left the hole injection occurs in the hole transport layer (HTL). On the right the electron injection takes place in the electron transport layer (ETL).The radiative recombination occurs in the organic emitter (pale yellow)
Similarly, electron injection from the ETL to the organic emitter and the electrons blocking at the HTL is ensured. In these way we increase the probability of exciton formation and radiative recombination in the emitting layer.
Organic Field Effect Transistor (OFET)
A transistor is a semiconductor device commonly used as an amplifier or an electrically controlled switch. The first field-effect transistor (FET) was inorganic and was invented in 1947 by John Bardeen, William Shockley and Walter Brittain (Nobel prize, 1956). Since their discovery, transistors have dominated the mainstream microelectronics industry; in fact, they are the fundamental building blocks for basic analytical circuits. An Organic Field-Effect Transistor (OFET) is a transistor based on organic semiconductors. The interest in using organic semiconductors as the active layers in FETs stemmed from the demonstration of field-effect conduction in small organic molecules [4] and conjugated polymers [4]. In 1986 the first OFET was reported by A. Tsumara [5]. From that moment a huge improvement in materials performances and development of new fabrication techniques took place.
OFETs are technologically attractive because all their layers can be deposited at low temperature and with low cost, in a large area and on a flexible substrate [6]. OFETs have also already been demonstrated in flexible electronic applications such as active matrix electronic paper displays, sensors, and low-cost radiofrequency identification cards (RFIDs). In order to render these devices more suitable for various applications an important step is to increase the charge carriers mobility. Indeed, the performances of OFETs are still lower with respect to those of the amorphous silicon devices. Since conjugated organic solid are more similar to insulators than semiconductors, charge transport in these materials is much less efficient than in conventional semiconductors. Clearly, the problem is more crucial in transistors where charges have to travel along much longer paths than in diodes. At the current state of the art, mobility in organic thin-film transistor ranges between 0.01 and 10 cm2/Vs, which is still much lower than what found in inorganic semiconductors (mobility is around 103 cm2/Vs in crystalline silicon), but substantially higher than the typical values obtained in organic light-emitting diodes or photovoltaic cells.
High mobility in OFET is the result of large research efforts at improving structural order in the organic semiconductor film. However, the physical origin of high mobility in organic solid is still an unresolved theoretical question. In addition to high mobility values, the major objectives are: stability under ambient conditions and under bias stress, device to device fabrication reproducibility as well as easy processing, e.g., from solution, which would make organic semiconductors a viable alternative to amorphous silicon.
OFETs working principles
A field-effect transistor is composed by a semiconducting material working as a channel in which current flows. At one extremity of the channel there is an electrode called source and at the opposite side there is a second electrode called drain. The physical dimension of the channel are fixed but the potion of the active material actually used for the conduction can be varied by applying a voltage to a third electrode called gate. The FET conductivity depends on the portion of the channel open to the current. Little changes in the gate voltage can involve great changes in the current flowing from the source to the drain, thus amplifying the signal. If the channel is composed by an organic material, we have an organic field-effect transistor (OFET). The channel is in contact with a dielectric layer working as a capacitor, and allows current modulation through the gate voltage The main constituting elements of an OFET are: three contacts (source, drain and gate), an active semiconducting material and a dielectric layer. These key elements can be differently combined to obtain different device structures. The most common configurations are: bottom gate - bottom contact (in which the drain and source electrodes are positioned directly on the dielectric film), bottom gate - top contact (in which the source and drain electrodes are grown on the organic semiconductor) and top gate - bottom contact (in which the dielectric film is deposited on the organic semiconductor and the gate contact is placed on top of it).
The bottom gate-top contact (BG-TC) configuration is illustrated in figure 3, to explain briefly how a classic OFET works. The physical dimension of the channel is fixed and it is described by the channel length (L) and the channel width (W). We refer to the voltage applied between drain and source as Vds, while the voltage applied to the gate is labelled as Vg. By convention, the source is generally considered grounded and the voltage is applied to the drain contact. The current flowing through the channel is called Ids and it is a strong function of Vg.
Figure 3. Classic BG-TC geometry with physical channel dimensions labelled: W as channel width, L as channel length.
The dielectric is sandwiched between the gate and the organic semiconductor, and they work as two plates of a plane capacitor. Thus, when a Vg is applied, charges of different sign are accumulated at gate/dielectric and dielectric/organic semiconductor interfaces (as in a classic capacitor, in Fig. 3 a positive gate voltage applied as an example). Much of this accumulated charge in the active material is mobile and moves in response to the applied Vds. When no Vg is applied there are ideally no free charge carriers, and the device is off. Otherwise, with applied Vg the device is on. This gate-induced charge carrier creation is called field-effect, and it is the key-idea of the working principle of FETs [7].
OFETs working principles
An n-channel OFET (i.e., electrons are transported through the channel) can be used as an example to understand the basic device operational regimes due to the gate voltage dependence of the distribution of “free” charges in the active material [8].
Real devices are far from being ideal and not all induced charges are mobile; a large number of deep charge traps are present in the film (deep enough to effectively immobilize electrons trapped in them). Deep traps have to be filled before the additionally induced charge can move. Thus, a minimum gate voltage has to be applied to obtain free electron density in the channel, the threshold voltage, Vt. Obviously, in n-channel OFETs Vt is higher than zero (for a p-channel OFETs, Vt is lower than zero since the charges flowing are positive).
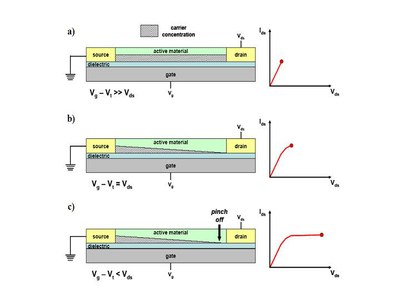
When a small source-drain voltage is applied (Vds<<Vg-Vt) a linear gradient of charge density is formed. The voltage drop between organic semiconductor and gate is larger at the source, where V(x) = 0 (grounded), than at the drain, where V(x) = Vds. This is the linear regime in which the current flowing through the channel is directly proportional to Vds (Fig. 4a).
Figure 4. Schematic structure of an OFET. a) Carrier concentration profile in the linear regime;
b) Carrier concentration profile when the pinch off occurs near the drain electrode (Vg – Vt = Vd);
c) Carrier concentration profile in the saturation regime.
When the source-drain voltage is further increased, we reach a point where a potential difference between the gate and the part of the channel near the drain no longer exists, the channel is pinched off. This means that a charge carrier depletion region is formed next to the drain, because the difference between the local potential V(x) and Vg is lower than the threshold voltage.
A space-charged-limited saturation current can flow across this narrow depletion zone. Carriers are swept from the pinch point to the drain by a comparatively high electric field in the depletion region (Fig.4b). A further increase in Vds pushes the pinch-off point further away from the drain (Fig. 4c). However the length of the channel (L) shortens only slightly, as it is infinitely larger than the width of the depletion region, and the integrated resistance of the channel from the source to the pinch point remains more or less the same. For these reasons once pinch off condition is met, current saturates at Idssat. From a mathematical point of view, pinch off is reached when Vds = Vg – Vt.
The current-voltage characteristics in the different operating regimes of an OFET can be described analytically, in a simplistic way assuming that (1) the transverse electric field induced by the gate voltage is largely higher than the longitudinal field induced by the gate bias (gradual channel approximation) and (2) the mobility is constant all over the channel.
OLET (Organic Light-Emitting Field-Effect Transitor)
While the development of ambipolar (both hole- and electron-transporting) OFETs is still in the early stages, it is certainly an exciting subject within the OFET community. These devices offer not only new possibilities for complementary logic circuit design, but also the potential to control electron-hole recombination within the semiconductor channel to afford light emission. Light-emitting organic field-effect transistors (OLETs) are particularly intriguing because they posses charge carriers densities that are order of magnitude higher than those found in OLEDs [9].
Even if different device architectures can be engineered for obtaining balanced ambipolarity and maximizing light emission, in Fig. 5 we report the scheme of single-layer and single-material BG-TC LET for showing LET main working principles and components. Indeed, a LET is a three-terminal device that couples the electrical characteristics of a FET to the controlled radiative recombination of the electrons and holes injected in the channel via the drain and source contacts. Excitons are thus created by the recombination of inplane moving electron- and hole-currents, which are controlled by the gate electrode. Electroluminescence intensity is tuned by both the drain and gate voltage. The gate electrode allows higher control on charge injection, electron and hole-currents balance and light-generation processes with respect to the standard sandwich configuration of an LED. Crucial aspects of the device’s characteristics concern (i) metal electrodes that should guarantee efficient electron and hole injection; (ii) active materials that should sustain ambipolar field-effect transport and efficient electroluminescence emission; (iii) dieletric surfaces that should be trap-free to favour effective mobility of both electrons and holes at the dielectric/active material interface
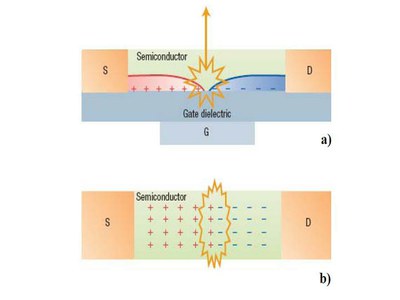
Figure 5. Scheme of a light-emitting field-effect transistor. (a) Side view. (b) Top view. In a simple model, the device can be modelled as a sort of a two-dimensional forwardbiased pn junction. Electrons and holes are injected from the drain (D) and source (S) contacts and recombine within the channel position controlled by the gate (G).
When an ambipolar FET is biased with the gate voltage in between the source and the drain voltage, an electron accumulation layer should form near one electrode coexisting with a hole accumulation layer near the other electrode. This regime can be difficult to reach even if electron and hole accumulation layers can be induced separately at high positive and negative gate voltages, respectively, because the coexistence of both carriers requires the threshold voltage difference for electron and hole accumulation to be sufficiently small. In the truly ambipolar regime, electrons and holes are expected to recombine where the two accumulation layers meet, leading to light emission from a welldefined zone. Based on energy band line-up considerations with the organic semiconductor HOMO and LUMO, depositing two different contact materials at either end of the transistor channel may facilitate more efficient hole and electron injection respectively. However, at the state of the art it is still unclear whether separately engineering distinct contacts for hole/electron injection in ambipolar OFET will prevail over opting for symmetric contacts.
Also in unipolar FET light-emission can occur [10], but radiative recombination is always confined to a spatial region near the drain metal electrode where the complementary charge carriers are injected.. Thus, in this thesis we will deal only with ambipolar OLET in which a gate-controlled location of the light emission is possible.
References
[1] P. R. Emtage, J. J. O’Dwyer, Phys. Rev. Lett., 1966, 16, 356.
[2] E. M. Conwell, M. W. Wu, Appl. Phys. Lett., 1997, 70, 1867.
[3] V. I. Arkhipov, E. V. Emelianova, Y. H. Tak, H. Bassler, J. Appl. Phys., 1998, 84, 848
[4] G. H. Heilmeier, L. A. Zanoni, J. Phys. Chem. Solids, 1964, 25, 603.
[5] A. Tsumara, H. Koezuka, T. Ando, Appl. Phys. Lett., 1986, 49, 1210.
[6] S. R. Forrest, Nature, 2004, 428, 911.
[7] C. R. Newman, C. D. Frisbie, D. A. da Silva Filho, J. L. Bredas, P. C. Ewbank, K. R. Mann, Chem. Mater., 2004, 16, 4436.
[8] J. Zamuseil, H. Sirringhaus, Chem. Rev., 2007, 107, 1296.
[9] J. Zaumseil, R. H. Friend and H. Sirringhaus, Nature Materials, 2006, 5, 69.
[10] A. Hepp, H. Heil, W. Weise, M. Ahles, R. Schmechel, H. von Seggern, Phys. Rev. Lett., 2003, 91, 157406.